Editor’s Notes

Total Results: 1697
Volume 107 : July 2022 Issue
Mineral evolution heralds a new era for mineralogy
https://doi.org/10.2138/am-2022-8414
"This Highlights and Breakthrough by Anhuai Lu is dedicated to the review article by Robert M. Hazen ""On the paragenetic modes of minerals: A mineral evolution perspective"" in this issue of American Mineralogist. As IMA President, Lu also wishes to use this contribution to congratulate Robert M. Hazen, who was honored with the IMA Medal for Excellence in Mineralogical Research 2020, for his outstanding achievements in mineral crystal chemistry, particularly in the field of mineral evolution."
Pauling’s rules for oxide-based minerals: A re-examination based on quantum mechanical constraints and modern applications of bond-valence theory to Earth materials
https://doi.org/10.2138/am-2021-7938
Recent criticisms of Pauling's Rules of crystal chemistry based on a statistical analysis of their applicability to some 5000 crystal structures have raised serious questions about their usefulness. Gibbs et al. have addressed these questions and concerns, showing that there is a firm quantum mechanical basis for these rules and that modification of the second rule to account for variation in bond strength with bond length has led to development of bond valence theory (BVT). They conclude by showing some of the many applications of BVT to Earth materials.
A cotunnite-type new high-pressure phase of Fe2S
https://doi.org/10.2138/am-2022-7959
Sulfur is frequently observed in iron meteorites and could be a common impurity element in planetary iron cores in our solar system and beyond. Therefore, the Fe-FeS phase diagram is of great importance to understand metallic cores of terrestrial planets. Here Oka et al. examined the pressure-induced phase transitions in Fe2S and discovered a new phase with the cotunnite-type structure that is stable between 30 and 130 GPa. It has been argued that sulfur is an important light element in the Mars' core, and they discuss the possible presence of cotunnite-type Fe2S there.
Density determination of liquid iron-nickel-sulfur at high pressure
https://doi.org/10.2138/am-2021-7924
The nature and the amount of light elements in the Earth's outer core has been one of the biggest enigmas of Earth sciences for more than 60 years. The comparison of seismic observations with sound velocity and density data measured in high pressure experiments can help reveal the lighter components in the Earth's outer core. Kawaguchi et al. determined the density of liquid (Fe,Ni)75S25 up to 74 GPa at 3400 K from pair distribution functions obtained by synchrotron X-ray diffraction measurements using diamond anvil cells. They obtained bulk modulus and its pressure derivative of liquid (Fe,Ni)75S25 using the third-order Vinet equation of state. The atomic volume change of Fe47Ni28S25 by melting was found to be approximately 10%, larger than that of pure Fe (~3%). They assumed that this large volume change was due to large anisotropy in the direction of the thermal vibrations of the Fe3S structure, based on the analysis of powder X-ray diffraction measurements at ambient pressure. Finally, assuming ideal mixing with pure Fe and S as the only light element in the core, the density profile of the outer core is best explained using 5.3-6.6 wt% S, an estimation compatible with sound velocity measurements, w.
On the paragenetic modes of minerals: A mineral evolution perspective
https://doi.org/10.2138/am-2022-8099
A systematic survey of the paragenetic modes of 5659 mineral species by Hazen and Morrison reveals patterns in the diversity and distribution of minerals related to their evolving formational environments. The earliest minerals in stellar, nebular, asteroid, and primitive Earth contexts were dominated by relatively abundant chemical elements, notably H, C, O, Mg, Al, Si, S, Ca, Ti, Cr, and Fe. Significant mineral diversification subsequently occurred via two main processes, first through gradual selection and concentration of rarer elements by fluid-rock interactions, and then through near-surface biologically-mediated oxidation and weathering. Most minerals form in only one way, though a few common minerals form by a dozen or more processes. A third of known mineral species form exclusively as a consequence of biological processes. By far the most significant factor in enhancing Earth's mineral diversity has been its dynamic hydrological cycle. At least 4583 minerals — 81 % of all species — arise through water-rock interactions.
Lumping and splitting: Toward a classification of mineral natural kinds
https://doi.org/10.2138/am-2022-8105
How does one best subdivide nature into kinds? All classification systems require rules for lumping similar objects into the same category, while splitting differing objects into separate categories. In the evolutionary system mineralogy Hazen et al. lump two minerals only if they have related composition, structure, and formation environment. They split two minerals if they form in two or more distinct paragenetic environments. Based on these lumping and splitting criteria, they estimate that Earth holds more than 10,000 mineral natural kinds.
Thermal expansion of minerals in the amphibole supergroup
https://doi.org/10.2138/am-2022-7988
This paper by Tribaudino et al. attempts to understand thermal expansion in the amphibole group of minerals. Such work will lead to a greater ability to predict phase equilibria that involve amphiboles at high temperatures, in this case those applicable to both metamorphic and igneous conditions. As a follow-on to their recent thermal expansion work on the pyroxene system, the data also allow further testing of various thermal expansion models.
A multi-faceted experimental study on the dynamic behavior of MgSiO3 glass in the Earth’s deep interior
https://doi.org/10.2138/am-2022-7969
Ryu et al. take a unique, multifaceted, experimental approach by studying MgSiO3 glass using three techniques at high pressure: Raman spectroscopy, X-ray scattering, and Brillouin spectroscopy. Their results present a complete and consistent picture of the structural modification of MgSiO3 glass under high pressure. A structural evolution model is proposed, explaining all the details of structural modification in MgSiO3 glass with pressure.
Origin of β-cristobalite in Libyan Desert Glass: The hottest naturally occurring silica polymorph?
https://doi.org/10.2138/am-2021-7922
Cavosie et al. report a geochemical and microstructural study of cristobalite, a silica polymorph in Libyan Desert Glass (LDG). LDG is a nearly pure silica glass found in Egypt and is thought to have formed during a meteorite impact. Our data reveal formation of oscillatory zoned beta-cristobalite at >1350 °C; during cooling it reverted to alpha-cristobalite. They propose that cristobalite in LDG represents the hottest naturally occurring silica polymorph thus far described, reflecting its unique formation during a meteorite impact.
Time-resolved Raman and luminescence spectroscopy of synthetic REE-doped hydroxylapatites and natural apatites
https://doi.org/10.2138/am-2022-8006
Fau et al. use time-resolved Raman and luminescence spectroscopies to characterize synthetic REE-doped hydroxylapatites and natural apatites. Using a unique experimental setup that allowswork at the microscale, they show how time-resolved spectroscopy can separate the Raman and luminescence signals for such samples. Such techniques is useful to study both signals without contribution/perturbation from the other signal. They discuss the possibility of semi-quantifying the REE concentration from luminescence spectra in such samples. Such quantification may be possible in samples doped with one single REE but is challenging in samples doped with several luminescence activators because of energy transfers between the various activators.
Reexamination of the structure of opal-A: A combined study of synchrotron X-ray diffraction and pair distribution function analysis
https://doi.org/10.2138/am-2022-8017
Opals (opal-A and opal-CT) are very common in young sedimentary rocks. New information about opal structures will help us better understand and quantity the transformation from opal-A (diatomaceous clay) to opal-CT during the diagenesis . The currently accepted opal structure is composed of disordered intergrowths of cristobalite- and tridymite-like domains consisting of six-membered rings of [SiO4] tetrahedra, but ILee et al. have identified additional, coesite-like domains comprising four-membered rings of [SiO4] tetrahedra. More broadly, this study has also demonstrated that the combined approach of synchrotron XRD/PDF is a powerful tool to determine the structures of poorly crystallized minerals. The combined method will provide the detailed crystallographic information of the phase transformation of natural opal.
A first-principles study of water in wadsleyite and ringwoodite: Implication for the 520 km discontinuity
https://doi.org/10.2138/am-2021-7929
Wang and Wu demonstrate that water is preferentially incorporated into wadsleyite relative to the coexisting ringwoodite and the presence of water enhances the formation of the 520-km discontinuity by influencing the two-phase coexistence domain.
Inclusions in calcite phantom crystals suggest role of clay minerals in dolomite formation
https://doi.org/10.2138/am-2020-7483
Farsang et al. characterized the micro- and nano-inclusions embedded in calcite phantom crystals from Gemerska Ves, Slovakia, by a combination of Raman spectroscopy, scanning and transmission electron microscopy, X-ray powder diffraction, and C and O isotope analysis. The inner, terracotta-colored part of the crystals contains abundant cavities, dolomite, hematite, goethite, titanite, phyllosilicate, and apatite inclusions and nanostructures that have grown on the walls of cavities: hematite and goethite particles sandwiched between either two phyllosilicate crystals or a phyllosilicate and a carbonate (calcite or dolomite) crystal. Their observations suggest that a smectite-illite conversion provides the Mg necessary for the precipitation of dolomite.
Crystal-chemical reinvestigation of probertite, CaNa[B5O7(OH)4]·3H2O, a mineral commodity of boron
https://doi.org/10.2138/am-2022-8086
Probertite, CaNa[B5O7(OH)4]x3H2O, is a mineral commodity of B (with B2O3~50 wt%). Its crystal chemistry was reinvestigated by Gatta et al. with a multi-methodological approach (X-ray and neutron diffraction, EPMA-WDS, LA-ICP-MS). Excluding B, probertite does not act as geochemical trap of other industrially-relevant elements (e.g., Li, Be, or REE). The H-bonding scheme in the structure of probertite is complex and pervasive, so that the H-bonding network presumably has an important role on the structural stability. The potential utilization of probertite is discussed.
Crystal structure determination of orthorhombic variscite2O and its derivative AlPO4 structure at high temperature
https://doi.org/10.2138/am-2022-8119
Variscite and metavariscite are uncommon secondary minerals, relevant in environmental and in technological applications. Although two orthorhombic modifications are known (Lucin- and Messbach-type), the fine-grained nature of the Messbach-type variscite has hampered determination of its crystal structure. Ardit et al. used XRD and NMR analyses to solve this conundrum. Structural interpretations and topological analysis demonstrate that the two orthorhombic modifications are polytypes. High-temperature XRD, NMR, and thermal analyses clarified the nature of the transformation to a new derivative AlPO4 structure.
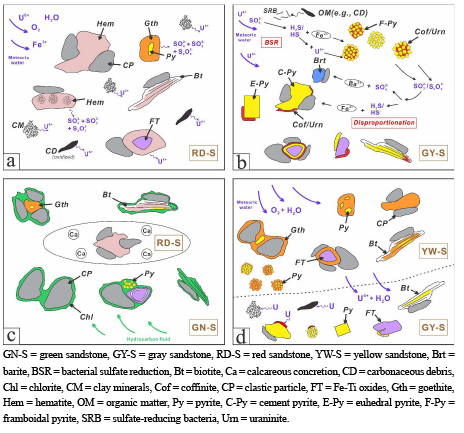
Transformation of Fe-bearing minerals from Dongsheng sandstone-type uranium deposit, Ordos Basin, north-central China: Implications for ore genesis
https://doi.org/10.2138/am-2021-7888
Yue et al. observe six main Fe-bearing minerals including biotite, Fe-Ti oxides, pyrite, hematite, goethite, and chlorite in four different colored sandstones. Mobilization and fixation of U are associated with alteration processes of Fe-bearing minerals. Paragenetic relationships and sulfur isotopic compositions of ore-stage pyrite suggest two distinct mechanisms for U mineralization.
Vaterite in a decrepitated diamond-bearing inclusion in zircon from a stromatic migmatite in the Chinese Sulu ultrahigh-pressure metamorphic belt
https://doi.org/10.2138/am-2021-7940
Vaterite occurs in a diamond-bearing, decrepitated inclusion of zircon from a stromatic migmatite in the Chinese Sulu UHP metamorphic terrain. Indigenous diamonds and amorphous C-Si-O and Zr-Si-O materials are associated with vaterite in this paper by Fei and Liu. The Raman spectra of highly disordered mcarbonaceous material in the amorphous materials indicate that it originated from organic matter. Inclusion decrepitation resulted in the precipitation of the amorphous materials and the formation and preservation of vaterite.
Oxygen diffusion in garnet: Experimental calibration and implications for timescales of metamorphic processes and retention of primary O isotopic signatures
https://doi.org/10.2138/am-2022-7970
Scicchitano et al. have uploaded a Graphical Abstract, to see online (http://www.minsocam.org/MSA/AmMin/AM_NotableArticles.html).
Oxidation state of iron and Fe-Mg partitioning between olivine and basaltic martian melts
https://doi.org/10.2138/am-2021-7682
Olivine dominates the upper mantle, is often the first phase to crystallize from a mantle melt, and has a relatively simple chemistry. As a result, it is critical for understanding and modeling igneous processe. Fe-Mg partitioning between olivine and melt can be used as a criterion of equilibrium in experiments and for the estimation of parental liquid compositions, which allows the calculation of mantle potential temperature, as well as putative mantle compositions capable of producing observed magmas. An important consideration is whether or not the canonical exchange partition coefficient, (Fe2+O/MgO)olivine/(Fe2+O/MgO)liquid or KD, value of 0.30 is appropriate for Martian applications as Martian melts have significantly more Fe and lower Al than their terrestrial counterparts. Calculating an accurate, KD, however, is difficult due to the fact that the silicate liquid has non-negligible amounts of both Fe2+O and Fe3+O1.5. To better constrain Fe3+/Fe2+ in magmatic liquids, and the corresponding KD of martian basalts, Matzen et al. conducted a series of experiments at controlled temperature and oxygen fugacity on a synthetic analog of a proposed primary Martian basalt. After quenching, Fe3+/Fe2+ ratios of the resultant glasses were determined by Mossbauer spectroscopy. They combined their data with experiments from the literature to determine the KD appropriate for Martian magmas. They used their KD to determine which, if any, of the olivine-bearing shergottites potentially represent liquids. For each meteorite, they assumed a liquid composition equal to that of the bulk, and then compared that liquid to the most Mg-rich olivine reported. Applying their KD, leads to the possibility that bulk Yamato 980459, NWA 5789, NWA 2990, Tissint, and EETA 79001 (lithology A) represent liquids.
Volume 107 : June 2022 Issue
Periodic and non-periodic stacking in molybdenite (MoSM2) revealed by STEM
https://doi.org/10.2138/am-2022-8019
Yang et al. used HAADF-STEM imaging to identify the stacking faults and domains of long-period molybdenite polytypes. Several layers of disordered domains intergrown with ordered 2H1 domain were recognized based on the contrast in HAADF image with one-dimensional lattice fringes. In addition, 30-layer long-period polytype and 2H3 polytype with three repetition domains were discovered for the first time. These results demonstrate that the presence of trace elements is strongly correlated with the existence of disordered domains in molybdenite crystals. It is suggested that non-equilibrium conditions related to compositional fluctuation of fluid during crystallization resulted in the oscillation of 2H1 and 3R polytypes and intergrowth of various disordered domains. The HAADF-STEM imaging method may be applied for studying other disordered layered crystals and twin minerals.
« ‹ 1 … 32 33 34 35 36 … 85 › »