Editor’s Notes

Total Results: 1697
Volume 98 : January 2013 Issue
Zircon growth and recrystallization during progressive metamorphism, Barrovian zones, Scotland
https://doi.org/10.2138/am.2013.4240
On page 219 of this issue, Vorhies et al. (2013) conduct SIMS U-Pb depth profiles of zircons in metamorphic rocks from the type section of Barrovian metamorphism in Scotland. Metamorphic grade for their samples ranges from the chlorite to sillimanite + K-feldspar. They find that even at the highest grades of metamorphism, only the very outermost margins of zircon rims (<1 micrometer depth) have their ages reset by metamorphism. Zircon interiors thus mostly record pre-metamorphic histories (pre-470 Ma), where in this case zircon interiors yield age dates of mostly 600 to 2000 Ma. In the outermost (<1 micrometer) zones, a range of age dates are recovered that reveal a complex history of decompression melting, later intrusion of granitic rocks, volcanism, and mineralization, ranging from 470 to 250 Ma. Only those zircon rims formed in the sillimanite zone contained rims that would be wide enough (>20 micrometers) to yield peak metamorphism age dates by spot analyses.
Editor's Notes Under Review
https://doi.org/10.2138/am-2022-8440
This paper by Wang et al. evaluates the controlling factors of hydrothermal wolframite and scheelite precipitation in the quartz-vein type Jiaoxi tungsten deposit (western part of the Lhasa terrane, Tibet, China) using texture, mineral major, and trace element geochemistry, and sulfur stable isotope geochemistry. Major and trace element contents and in-situ S isotope compositions of pyrite and chalcopyrite show that an early magmatic fluid which is characterized by the enrichment of W, Mn, Zr, Ti, Sc, and Sn and the depletion of Fe. This magmatic fluid mixed with meteoric water inducing dilution and interacted with the biotite monzogranite porphyry to leach Fe, Mg, and Zn into the system to form wolframites with variable Fe/(Fe+Mn) ratios. The late, Fe-enriched magmatic fluid released from the muscovite granite mixed with meteoric water that leached minor Fe and S from shale to form the Fe-enriched wolframite The compositional zoning of wolframite and scheelite grains as revealed by trace element mapping results indicate a pulsatory magmatic fluid. This study highlights that multiple Fe sources were present in the quartz vein-type tungsten deposit hydrothermal system.
https://doi.org/10.2138/am-2023-9122
The Fe oxidation state of minerals is an important constraint for the formation and evolution of rocks. This new software developed by Hezel et al. allows other laboratories to quickly adopt the flank method to determine mineral Fe oxidation states. Determining the oxidation state of a mineral can be challenging as a specific method such as Mossbauer or XANES might not be available or has an insufficient spatial resolution. The flank method offers an elegant way to determine the oxidation state of a mineral highly localized using an electron microprobe. A key challenge of this method was the data reduction process, which was complex and time-consuming. This software is a freely available web application with a simple data process that offers additional functionalities to explore and test the data.
https://doi.org/https://doi.org/10.2138/am-2022-8735
Small amounts of water (10s to 100s of ppm) are often present in samples derived from the Earth's mantle. Water can profoundly affect the properties of mantle peridotites, including viscosities, conductivities, and melting temperatures. Measuring the water content of nominally anhydrous minerals (NAMs) has provided insight into the amounts of water contained within mantle rocks. However, converting from NAM water contents to the activity of H2O, a(H2O), is non-trivial. Equilibria involving amphibole can be used to determine values of the a(H2O) at the time of mineral equilibration. This approach yields low values of a(H2O) (<0.3) for four peridotite xenoliths from Southeastern Australia. These four xenoliths also record oxygen fugacity values ranging from -0.2 to -1.2 (Log units relative to FMQ). All these values of oxygen fugacity are inconsistent with the presence of a CH4-rich fluid (too oxidizing), and the lowest value of oxygen fugacity, as recorded by one sample, is inconsistent with the presence of a CO2-rich fluid.
https://doi.org/https://doi.org/10.2138/am-2023-9059
The apatite mineral group contains anions of chlorine and OH, allowing insight into the behavior of geologic fluids. Recent crystal structural refinements found new anionic positions along the c-axis. Polarized infrared spectroscopy of single crystals found changes compared to the hydroxyapatite end-member. A pair of OH anions, spaced 2.9 apart, is the only contributor to the hydrogen-bonded spectra. In crystallographically similar positions, an OH-Cl pair appears to shift the OH stretching frequency to lower wavenumbers, indicative of hydrogen bonding. However, the interatomic distance is too great to allow hydrogen bonding. The structural changes that mimic hydrogen bonding are elucidated. Another peak in the infrared is shifted to higher wavenumbers in the OH stretching domain, the opposite of hydrogen bonding. This peak results from a chlorine atom interacting with the oxygen end of the OH dipole. The chlorine-oxygen distance for the pair is very short, and the means of accommodation are discussed.
https://doi.org/https://doi.org/10.2138/am-2023-9091
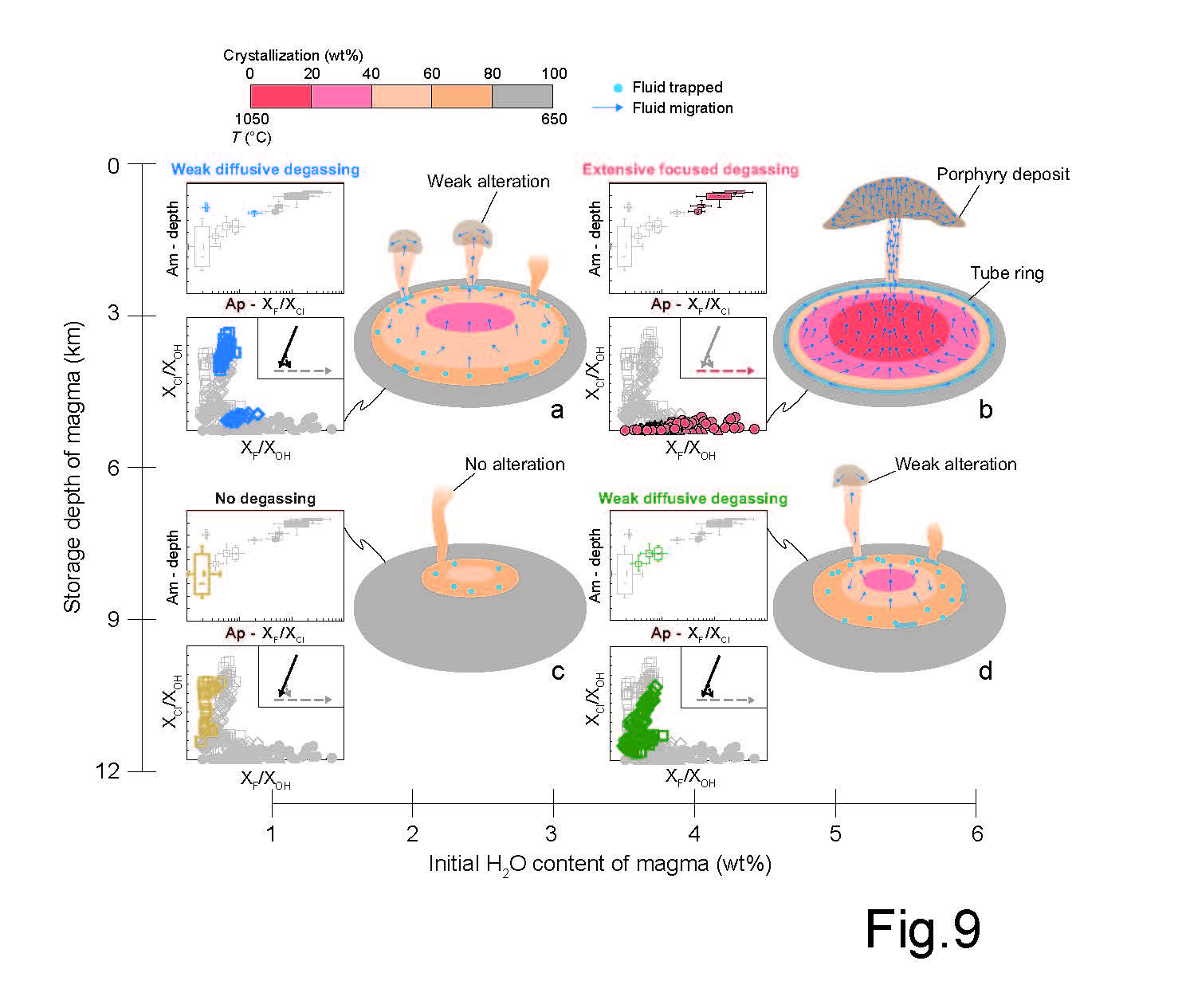
Metals, such as copper and gold, play important roles in human life. However, they have relatively low contents within the Earth's interior and require focusing processes at shallow depths to form deposits (such as porphyry) before they can be exploited and utilized. In this study, porphyry deposits are taken as the research objects. The behaviors of magmatic fluid, closely related to the metal migration process at different depths in the shallow crust, are characterized by the comprehensive application of geochemistry, mineralogy, and thermodynamic modeling. Our results demonstrate that magma with shallower storage depth and higher initial H2O content is more likely to experience extensive focusing processes of magmatic fluid, leading to the generation of porphyry deposits.
https://doi.org/https://doi.org/10.2138/am-2023-9092
This study introduces Òdecision mapsÓ, a novel visualization technique for machine learning, to classify mineral genetic types using pyrite trace elements. This method enhances the interpretability of complex geochemical data, bridging the gap between traditional discriminant diagrams and modern machine learning efficiency. The approach provides a more transparent and understandable classification of mineral genesis, aiding geoscientists in better understanding the relationship between geo-data and decision-making in mineral exploration. This advancement marks a significant shift in geochemistry classification towards a visually insightful methodology.
https://doi.org/https://doi.org/10.2138/am-2023-9140
Raman elastic geothermobarometry is based on the determination of strain recorded by a mineral inclusion trapped in its host to estimate the entrapment pressure and temperature conditions of the inclusion. Determining the entrapment conditions of mineral host-inclusion systems in metamorphic rocks allows us to better understand the rock history and the geological processes. Raman elastic geobarometry has been successfully applied to anisotropic inclusions, such as quartz and zircon, entrapped in isotropic (i.e., garnets) and anisotropic hosts. Here, Baratelli et al. have extended the application of Raman elastic geobarometry to omphacite-in-garnet to develop a complementary system to the commonly used quartz or zircon inclusion in garnets. The authors report the pressure dependence of Raman spectra of omphacite crystals with the same composition but different symmetry because of the presence (P2/n) or absence (C2/c) of chemical order at the octahedral sites. They carried out ab initio Hartree-Fock/Density Functional Theory simulations on fully ordered omphacite to complement the experimental results. The results show that the changes in the position of the well-resolved Raman peak near 680 cm-1 with pressure are not affected by the presence or absence of chemical order, thus making it a good candidate for use in Raman geobarometry.
https://doi.org/https://doi.org/10.2138/am-2023-9150
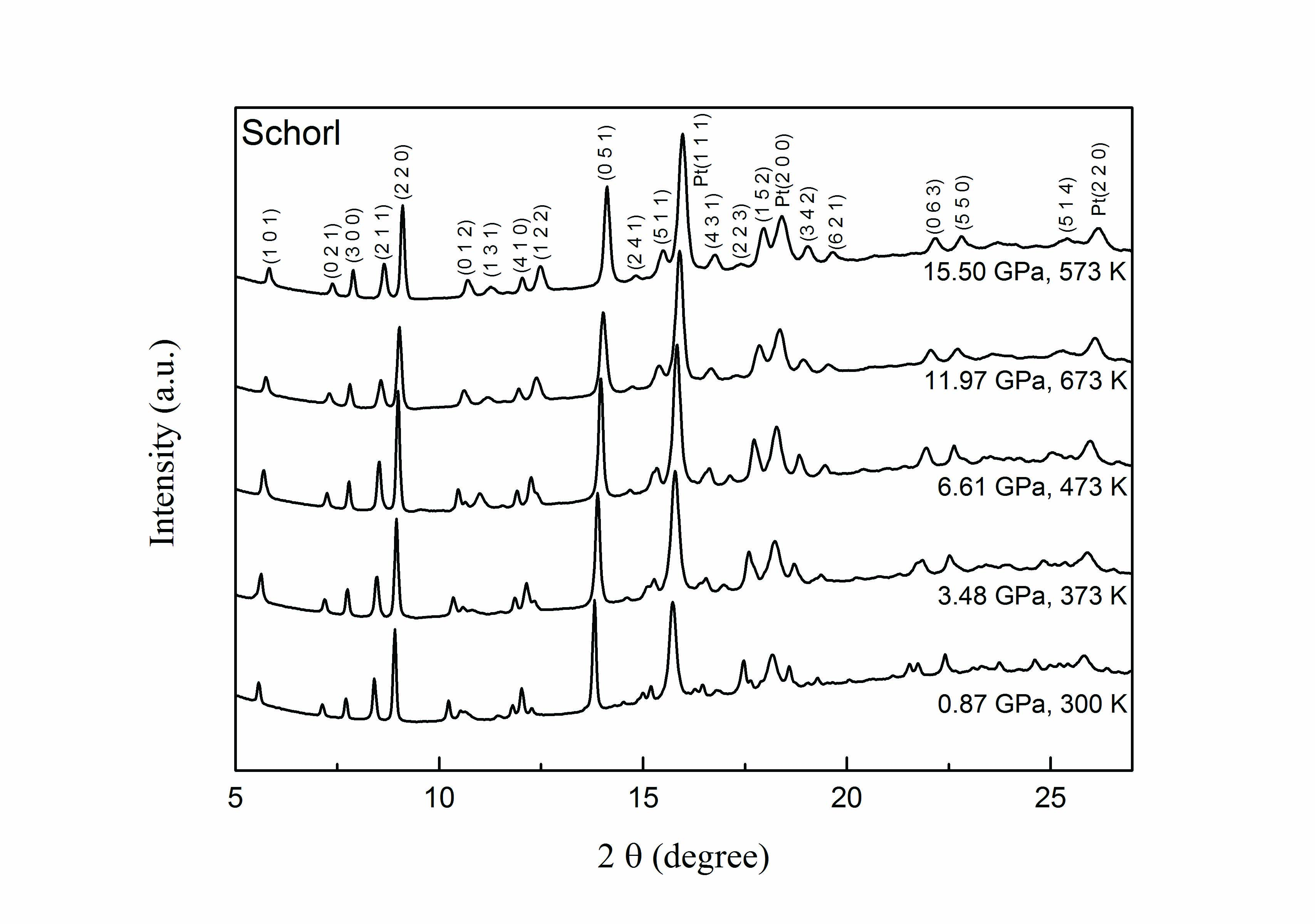
Tourmaline is the most widespread borosilicate mineral with a broad chemical composition. It occurs in a wide variety of granites, granitic pegmatites, and sedimentary and metamorphic rocks. As a dominant carrier of light elements, tourmaline plays a vital role in the lithium and boron cycle in the deep Earth, especially in the subduction zone. In subduction zones, as the pressure and temperature of the slab increase, the decomposition of lithium-bearing tourmaline will release fluxing elements like lithium and fluorine, which can induce partial melting and form lithium-rich magmatism. Thus, the thermal stability and equation of state (EoS) studies of lithium-bearing minerals such as tourmaline in the subduction slab help assess lithium cycle depth and explain the formation of lithium deposits. Pressure-volume EoS studies of tourmalines have been widely carried out using synchrotron X-ray diffraction combined with a diamond-anvil cell. In this study, Chen et al. investigated the pressure-volume-temperature relations of Li-rich schorl at high pressures up to 15.5 GPa and high temperatures up to 673 K. The compression property of schorl at room-temperature and high-pressure conditions was obtained. In addition, the authors compared the compressibility of schorl and elbaite and analyzed the possible factors influencing it. The anisotropic linear compressibility of schorl and elbaite was also discussed. Finally, the thermal EoS properties of schorl are obtained by fitting the pressure-volume-temperature data to the high-temperature Birch-Murnaghan EoS.
https://doi.org/https://doi.org/10.2138/am-2023-9157
Johnson et al. have developed a protocol to analyze trace amounts of sulfur down to 17 ppm on the electron probe microanalyzer (EPMA) by applying a beam current of 200 nA and a total counting time of 200 s to a suite of basaltic glasses. They used this protocol to analyze S in silicate minerals in experimental assemblages bearing silicate glass + clinopyroxene ± garnet ± sulfide and found that the nominally S-free minerals bear up to ~50 ppm S. This represents the first measurement of S in experimental garnets at sulfide-saturation. They calculated mineral-melt partition coefficients for S of up to 0.110, indicating that S partitions are incompatible with silicate minerals, which agrees with earlier work.
https://doi.org/https://doi.org/10.2138/am-2023-9208
The authors studied the high-pressure behavior of Mg2GeO4 olivine, a commonly used analog of forsterite, Mg2SiO4. They show the presence of a high-pressure orthorhombic phase under room-temperature compression and a partial dissociation into bridgmanite and MgO or post-perovskite and MgO on laser heating. Using static and dynamic compression techniques, the high-pressure phase has now been reported in both silicates and germinates. This may have important implications in planetary interiors where it may persist as a metastable phase, thereby affecting the dynamics and structure of these planets.
https://doi.org/https://doi.org/10.2138/am-2023-9225
Inclusions of varying mineralogy are relatively common in certain types of iron meteorites. Herd et al. describe three new iron phosphate minerals from the 15-t El Ali iron meteorite found in Somalia. Discovered during the investigation and classification of the meteorite, the minerals are iron phosphates elaliite and elkinstantonite and K-bearing olsenite. Elkinstantonite is ferrous-iron dominant, whereas elaliite has mixed iron valency. Olsenite is the potassium analog of galileiite. The three minerals occur alongside wŸstite, troilite, and sarcopside/graftonite in inclusions ranging from 20 to over 200 _m across; larger inclusions have more varied mineralogy. These mineral assemblages reflect local chemical and redox conditions of their micro-environments, distinct from those found in other iron meteorites, although the minerals likely exist in other meteorites and have yet to be identified.
https://doi.org/https://doi.org/10.2138/am-2023-9240
Inorganic X-ray amorphous materials (iXAMs) are ubiquitous in soils but difficult to identify and quantify. Akinbodunse et al. evaluated the Rietveld method for quantifying iXAMs (ferrihydrite and opal-A) in crystalline mineral mixtures and determining their chemical composition based on Rietveld and chemical analysis data. The results provide a methodological framework for studying iXAM abundances and chemical compositions in soils and investigate their impact on soil physicochemical properties and biogeochemical element cycles.
https://doi.org/https://doi.org/10.2138/am-2023-9254
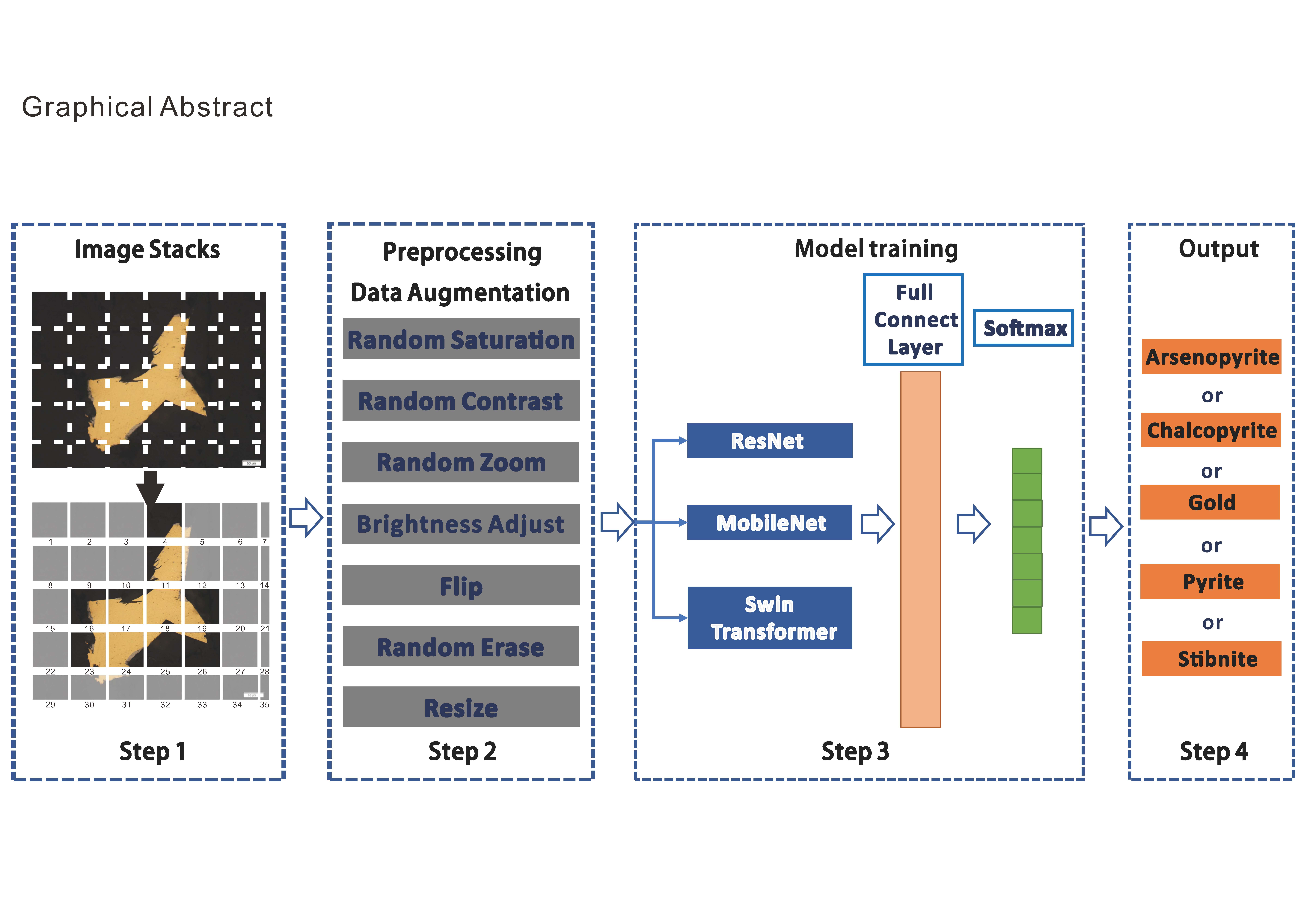
The analysis of optical microscopic image data is crucial for the identification of mineral phases. As such images are typically very large and difficult to collect, their analysis has a high computational (and often manual) cost and a limited amount of automation. Here, Wang et al. adopt the "transfer learning" paradigm. By testing the Swin Transformer, a deep learning algorithm for different metallic mineral phases, they propose a well-behaved Swin Transformer metal mineral classifier with a high accuracy of 92% and a wide application potential. Moreover, the results demonstrate that pre-training weights can enhance learning and capture the relevant attributes in mineral classification. However, deep learning approaches are often considered a black box in that they may be too complex to be easily interpretable by humans. The authors use the Class Activation Map for the first time for mineral classification tasks, and observe that mineral edges are the most effective model classification features. The results demonstrate that boosting the learning processes with pre-trained weights can accurately capture relevant attributes in mineral classification, revealing the potential for application in mineralogy and petrology and enabling its use in resource explorations.
https://doi.org/https://doi.org/10.2138/am-2023-9273
In unraveling the geological complexity of Luobusa Chromitite, this study investigates the features of Luobusa chromitite and ophiolites, revealing a multi-stage genesis involving mantle melting, melt-rock interactions, and shifts in oxygen and sulfur fugacity. Abundant platinum-group minerals, base-metal sulfides, and PGE-sulfides/alloys in chromitite signify intricate processes. Geochemical analysis and sulfur isotopes trace the Luobusa ophiolite's evolution from sub-ridge to supra-subduction zone settings. Variable _PGE values in chromitite suggest extensive melt fractionation. Well-crystalline Os-Ir alloys indicate early high-temperature stages, while sulfarsenide and pyrite inclusions show later stages with lower temperature and higher sulfur fugacity. Pyrite isotopes resonate with MORB and OIB characteristics, showcasing diverse magmatic sources.
https://doi.org/https://doi.org/10.2138/am-2024-9331
The authors establish an empirical equation giving the apatite etch rate as a function of orientation. This allows them to calculate the geometries, effective etch times, and etch rates of fission tracks across different chemistries. An etch-rate adapted protocol gives confined fission tracks with more comparable properties in apatites with different chemistries. Measuring the etch rates of fission tracks exhibits variation between apatites that could be related to their thermal histories.
https://doi.org/https://doi.org/10.2138/am-2024-9343
Intervalence charge transfer is a dominant cause of color in many minerals containing iron in mixed oxidation states or Fe2+ with Ti4+. This work examines corundum and aluminosilicates with these cations and shows that the color is greatly reduced or disappears at elevated temperatures. This work raises the possibility that the optical transmission of minerals at elevated deep Earth temperatures can radically differ from the room-temperature properties, with implications for radiative thermal transport in the Earth.
« ‹ 1 … 83 84 85